|
The Prescription Drug User Fee Act (PDUFA) action date is July 7, 2022. An updated label would allow the Company’s commercial team to promote KRYSTEXXA plus methotrexate to physicians. 
The Company submitted an sBLA in January based on results from the MIRROR Phase 4 randomized placebo-controlled trial which demonstrated that 71% of patients receiving KRYSTEXXA plus methotrexate achieved a complete response, a more than 30 percentage-point improvement compared to patients who were randomized to receive KRYSTEXXA plus placebo (p<0.0001). Food and Drug Administration (FDA) granted priority review of the Company’s sBLA to expand the label for KRYSTEXXA to include co-treatment with methotrexate. FDA Granted Priority Review of sBLA for Co-Treatment of KRYSTEXXA Plus Methotrexate: In March, the U.S.government-mandated COVID 19-vaccine orders.įirst Quarter and Recent Company Highlights 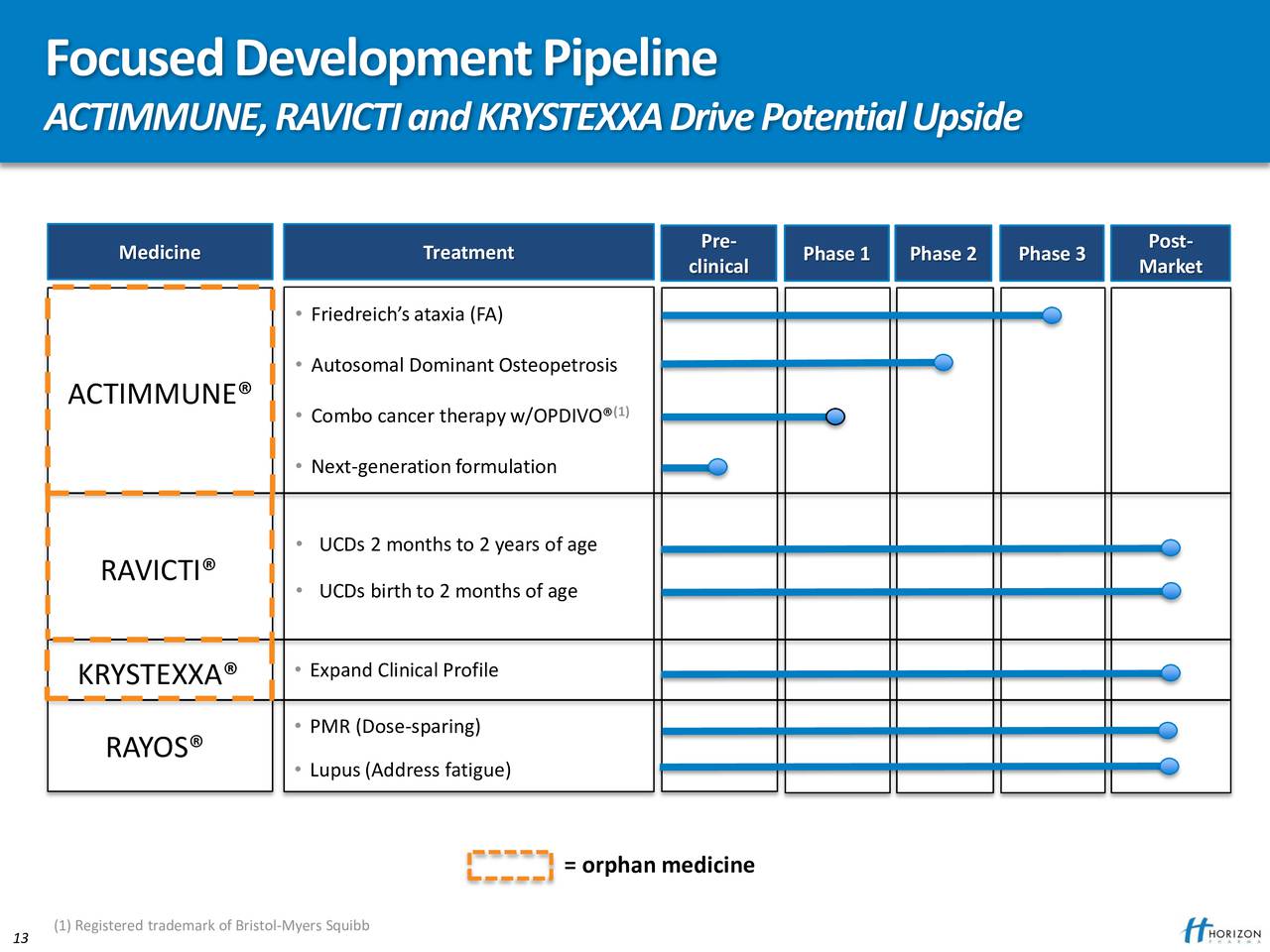
Announced Positive Topline Data from Phase 2 Trial Evaluating Dazodalibep (HZN-4920) in Patients with Rheumatoid Arthritis (RA) Study Met Primary Endpoint and Dazodalibep was Well Tolerated -įirst-quarter 2021 results were negatively impacted by a short-term TEPEZZA supply disruption due to U.S. Initiated Two Clinical Trials to Date, Five Additional Trials Expected to Initiate this Year. Initiated Launch Preparations to Support Potential Approvals for TEPEZZA and UPLIZNA in Brazil as part of Global Expansion Strategy. European Commission Approved UPLIZNA ® (inebilizumab-cdon) for the Treatment of Adult Patients with NMOSD. FDA Granted Priority Review of the Company’s Supplemental Biologics License Application (sBLA) for Co-Treatment of KRYSTEXXA Plus Methotrexate with a JPDUFA Action Date. Continue to Expect KRYSTEXXA Net Sales Growth of More Than 20%. Continue to Expect TEPEZZA Net Sales Percentage Growth in the Mid-30s. Maintaining Full-Year 2022 Adjusted EBITDA Guidance of $1.63 Billion to $1.70 Billion, Representing 30% Growth and 230 Basis Points of Margin Expansion at the Midpoint. Maintaining Full-Year 2022 Net Sales Guidance of $3.9 Billion to $4.0 Billion, Representing 22% Growth at the Midpoint. Cash Position of $1.64 Billion as of Ma. KRYSTEXXA ® (pegloticase injection) Net Sales of $140.7 Million. TEPEZZA ® (teprotumumab-trbw) Net Sales of $501.5 Million. GAAP Net Income of $204.3 Million Adjusted EBITDA of $371.2 Million.
0 Comments
Initially, the ICON ship design was announced with passenger capacity ~5200 and GT tonnage ~200K. For comparison, the previously largest passenger ships (OASIS PLUS class) have GT ~227000 tons and max capacity 8800 (6410 pax + 2390 crew). On October 20th, RCI revealed more details, including the vessel's exact volume (250800 GT-tons) and max capacity (9950/7600 passengers + 2350 crew). On May 5, 2022, an RCI spokesperson announced that the ICON class will be bigger (as both passenger capacity and volume) than the OASIS class, which makes Icon of the Seas currently the world's largest passenger ship ever constructed. RCI also has RCG's largest fleet by both vessel number and GT tonnage (ship sizes and capacities).Īll RCI liners have names ending with "of the Seas".Įach vessel from the ICON series has passenger capacity 5610 (lower berths) and GT tonnage/volume 250800 tons, making the design Royal Caribbean's/world's largest. RCI was established in 1968 and currently has over 1/5 of the worldwide cruise market. Royal Caribbean International (RCI) is a US company, trademarked brand, and subsidiary owned by Royal Caribbean Group (RCG/shipowner). The vessel (IMO number 9829930) is currently Bahamas- flagged (MMSI 311001178) and registered in Nassau. "Project Icon" is the name for a passenger ship class ordered by RCG-Royal Caribbean Group/shipowner to Meyer Merf's Shipyard in Turku Finland. The liner is currently the world's largest by volume/GT-tons (~6% bigger than Wonder OTS) and has two same-designed sisterships - Apex Of The Seas (2025-Q2) and Joy of the Seas (2026-Q2). The 2024-built Icon of the Seas cruise ship is RCI-Royal Caribbean's first ICON-Class vessel scheduled for delivery and inauguration in 2024 January, being postponed from 2022-Q-Q3 due to the COVID crisis. 7 days, round-trip Eastern Caribbean Perfect Dayħ days, round-trip Western Caribbean Perfect Day These come in different kinds such as two-photo collages, four-photo collages, top-text, bottom text, text on every image in the collage, all these meme template options are editable. Crello Free Meme Maker Crello FREE Meme maker websiteĬrello meme maker website is one of the many meme making websites which have a clean interface, and make creating memes easy.Īt first sight, you are able to select from the available templates. > Include top text, bottom text, meme title and Make the meme 3. > You will be prompted to upload an image as shown above > Click on make a meme and save the meme image How to create a custom meme with the make a meme website Make a custom meme with the ‘make a meme’ website > Scroll down for other options like removing the watermark > You can choose to make the meme public or private > Select a meme picture from the available options / Blank meme generator > Ensure to login/register on the website How to create/edit a meme with the ‘make a meme’ website Select a meme Edit a meme How to edit/make a meme with the ‘make a meme’ website
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |




 RSS Feed
RSS Feed
